- Home
- About Us
- Work
- Journal
- Contact
- Car loan calculator
- Gibson thunderbird bass
- Idle champions of the forgotten realms blessings
- Channel 8 news anchor fired
- Marvel ultimate alliance pc max settings
- Need for speed underground 2 walkthrough
- Magix photostory help
- Venture forthe director of nursing
- Serial cloner 2-6-1 free download
- Auto ac compressor repair
- Potassium atom
- Utorrent client
- Jquery cycle through nasa picture of the day
- Culcah candela
- Flamingo pink
- Best linux system monitor remote prettiest
- Nostalgia cotton candy machine
- Vernissage lyrics english
- Green river bible church co
- Nvidia container toolkit
- Hip hop art
- Home
- About Us
- Work
- Journal
- Contact
- Car loan calculator
- Gibson thunderbird bass
- Idle champions of the forgotten realms blessings
- Channel 8 news anchor fired
- Marvel ultimate alliance pc max settings
- Need for speed underground 2 walkthrough
- Magix photostory help
- Venture forthe director of nursing
- Serial cloner 2-6-1 free download
- Auto ac compressor repair
- Potassium atom
- Utorrent client
- Jquery cycle through nasa picture of the day
- Culcah candela
- Flamingo pink
- Best linux system monitor remote prettiest
- Nostalgia cotton candy machine
- Vernissage lyrics english
- Green river bible church co
- Nvidia container toolkit
- Hip hop art

#Potassium atom portable
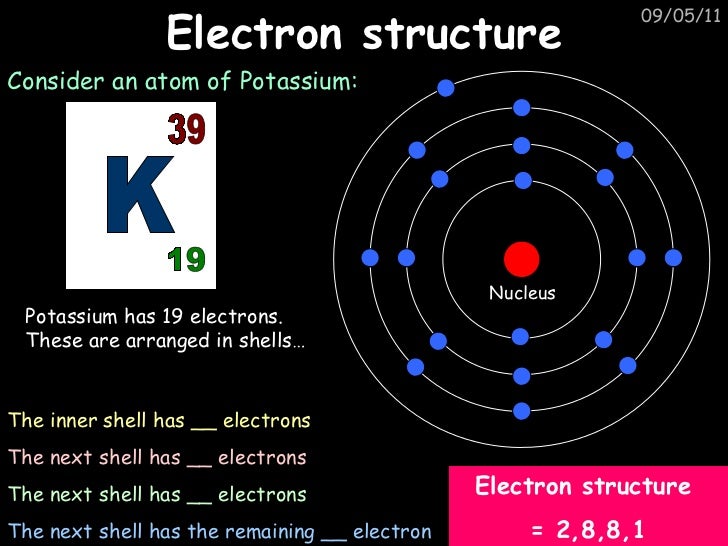
The melting point of potassium is 63.4 degrees C or 336.5 degrees K and its boiling point is 765.6 degrees C or 1038.7 degrees K.Potassium metal has a density of 0.89 grams per cubic centimeter.The atomic weight of potassium is 39.0983.The most abundant isotope is K-39, which accounts for 93.3% of the element. Three isotopes of potassium occur naturally on Earth, although at least 29 isotopes have been identified.Potassium is the second lightest (least dense) metal after lithium.Element number 19 is the eighth most abundant element in the human body, accounting for between 0.20% and 0.35% of body mass.Potassium is the seventh most abundant element in the Earth's crust, accounting for about 2.5% of its mass.Potash and alkali are two of the potassium compounds known to man since ancient times.

The symbol for potassium is K, which is derived from the Latin kalium and Arabic qali for alkali.

Potassium was the first metal to be isolated using electrolysis.
#Potassium atom free
Because of its high reactivity, potassium is not found free in nature.Potassium is one of the alkali metals, which means it is a highly reactive metal with a valence of 1.This means the atomic number of potassium is 19 and each potassium atom has 19 protons. Appearance: Potassium is a solid, silvery-gray metal at room temperature.
